Comprehensive Solutions for Extracellular Vesicle Production
Empower Your EV-Based Therapies With Solutions Driven by Reproducibility, Scalability, and Quality
Extracellular vesicles (EVs), also referred to as exosomes, are nano-sized particles with high biocompatibility, ideal for delivering genetic material and therapeutics. While they have high potential for use in oncology, regenerative medicine, and diagnostics, EV production is hampered by challenges in yield, purity, scalability, and characterization. Our end-to-end solutions ensure reproducibility, scalability, and quality — backed by decades of expertise and global accessibility.
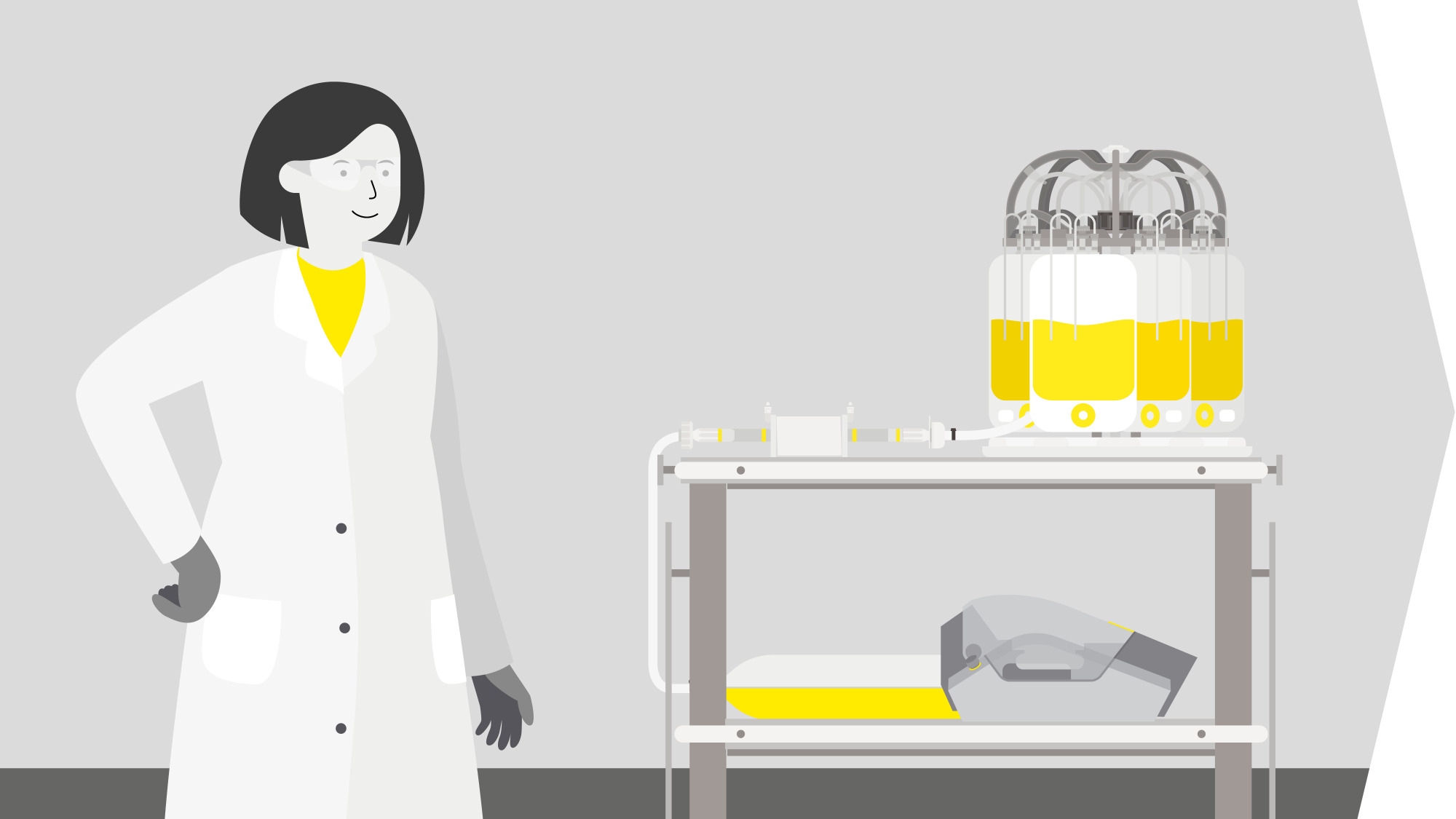
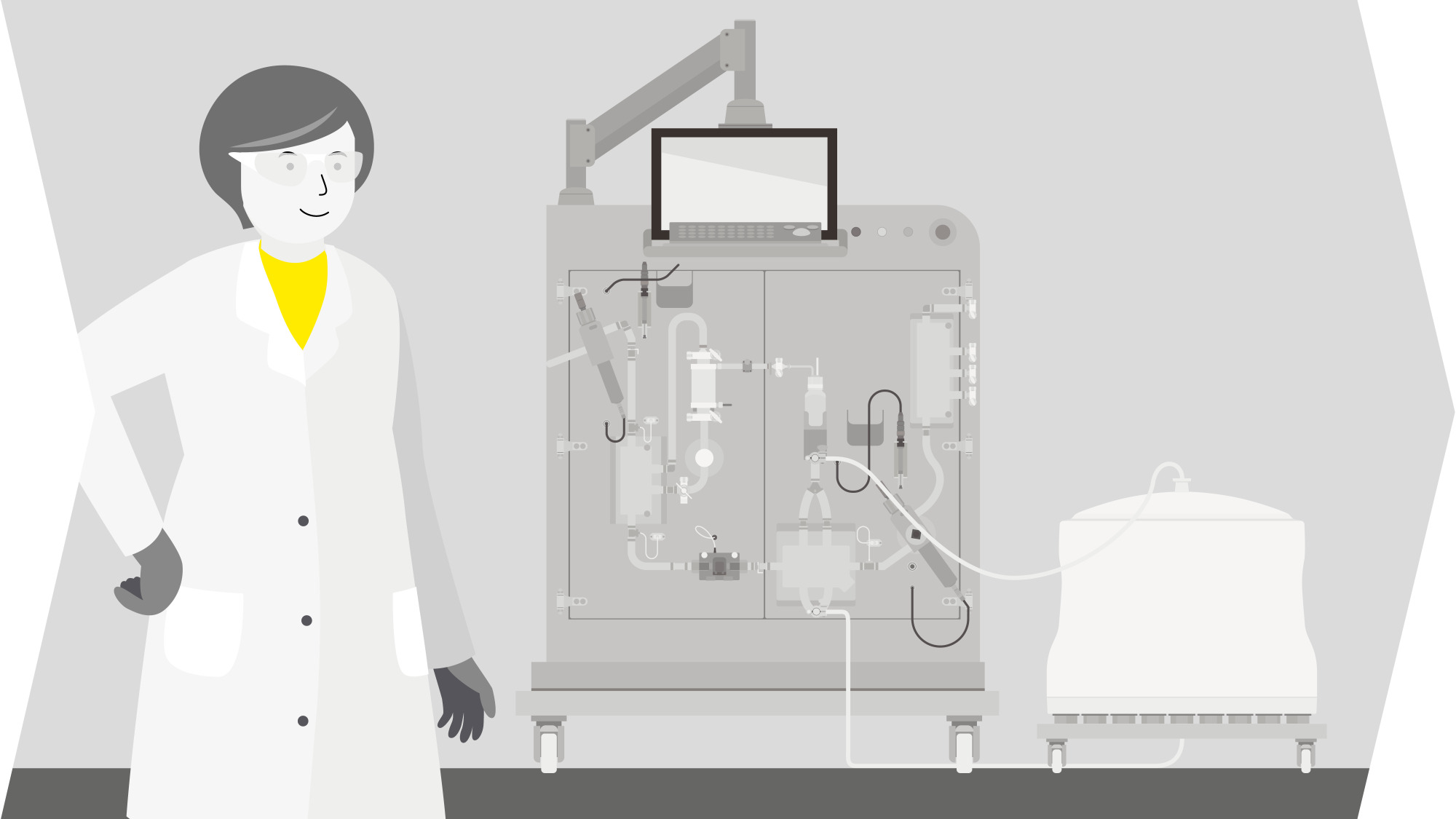
EV-Based Therapy Workflow
Solutions for EV-Based Therapies
Connected Services
Our process development service simplifies your path to the clinic for cell and gene therapies. Expert support and proven Sartorius technologies help scale processes, reduce variability, and accelerate time-to-market.
Comprehensive analysis of fresh and spent media to confidently monitor the impact of media components and process changes, enabling smarter optimization and more consistent process performance.
Our biologics testing services support product safety, quality, and regulatory compliance from development through commercial manufacturing. cGMP‑compliant testing and expert assays from Sartorius help de‑risk product release, streamline timelines, and accelerate your path to market.
Connected Software & Analytics
DOE software Simplified for Life Science. Ready-to-go connectors for Sartorius equipment & Biobrain Supervise to ease implementation. Backed up with a data science team to get the most out of your data.
Multivariate data analysis software that simplifies your data exploration for Process Experts. Ready-to-go connectors for Sartorius equipment and Biobrain Supervise. Backed up with a data science team to get the most out of your data.
A data acquisition, process control & monitoring system that connects Sartorius and 3rd party unit operations. A scalable system can support 1 to 32 process units to enable PD or GMP production with integration to Umetrics® for DOE or advanced process control.
Request Your Sample | Raw Materials and Consumables
To help find the right materials and consumables for your needs, request a sample from our portfolio.
More About EV-Based Therapies From Sartorius
Related Assets
FAQs for Exosomes
Exosomes are small, membrane-bound vesicles released by cells into their surrounding environment. They range from 30 to 150 nanometers in diameter and carry proteins, lipids, and nucleic acids that can mediate communication between cells.
Extracellular vesicles (EVs) are a broad category of membrane-bound particles released by cells, which includes exosomes as one subtype. Exosomes are specifically small EVs formed inside the cell and released when multi-vesicular bodies fuse with the cell membrane.
Exosomes are used in research and medicine for cell-to-cell communication studies, as biomarkers for disease diagnosis, and as vehicles for drug and gene delivery. Their natural cargo and targeting abilities make them promising for therapeutic and diagnostic applications.
Exosome therapy refers to the use of purified exosomes to promote healing, reduce inflammation, or deliver therapeutic molecules to specific cells or tissues. This emerging treatment approach is being studied for its potential in regenerative medicine and immune modulation.
Exosomes are tiny vesicles released by cells, while stem cells are living cells capable of self-renewal and differentiation into various cell types. Exosomes can be produced by stem cells and may mediate many of their therapeutic effects, but exosome-based therapies do not involve transplantation of live cells.
Benefits of exosome-based therapies include lower risk of immune rejection, ability to cross biological barriers, targeted delivery of therapeutic cargo, and potential for less invasive administration compared to cell-based therapies.
Challenges include efficiently isolating and purifying exosomes, ensuring consistency and safety in therapeutic preparations, scaling up production for clinical use, and understanding the full range of biological activities and potential side effects.
The three main types of extracellular vesicles are: exosomes (30–150 nm, formed within endosomal compartments), microvesicles (100–1000 nm, bud directly from the plasma membrane), and apoptotic bodies (500–2000 nm, released during programmed cell death).