Comprehensive Lentivirus Process Solutions
Setting The Standard in Lentiviral Solutions for Gene Therapy and Gene-Modified Cell Therapy, Together
Scientists often use lentiviral (LV) vectors to deliver genes to therapeutic cells (gene-modified cell therapies) and offer potentially life-saving treatment against cancers or delivered directly to patients to treat rare genetic disorders. Because developers can use LV as a raw material or as the final therapy product, it’s critical to build robust processes that achieve consistently high viral yields with high quality.
Lentiviral Vector Gene Therapy Workflow
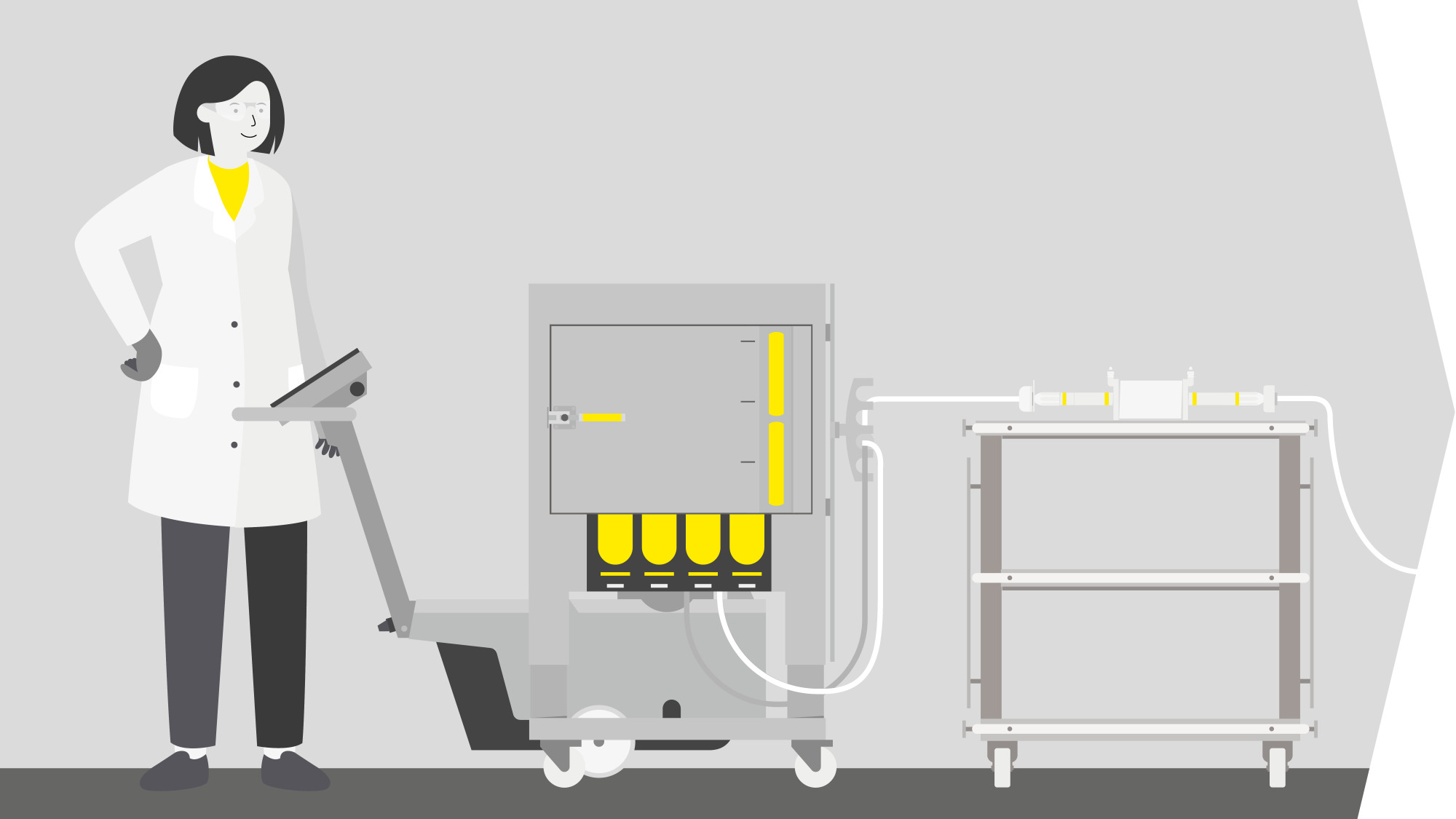
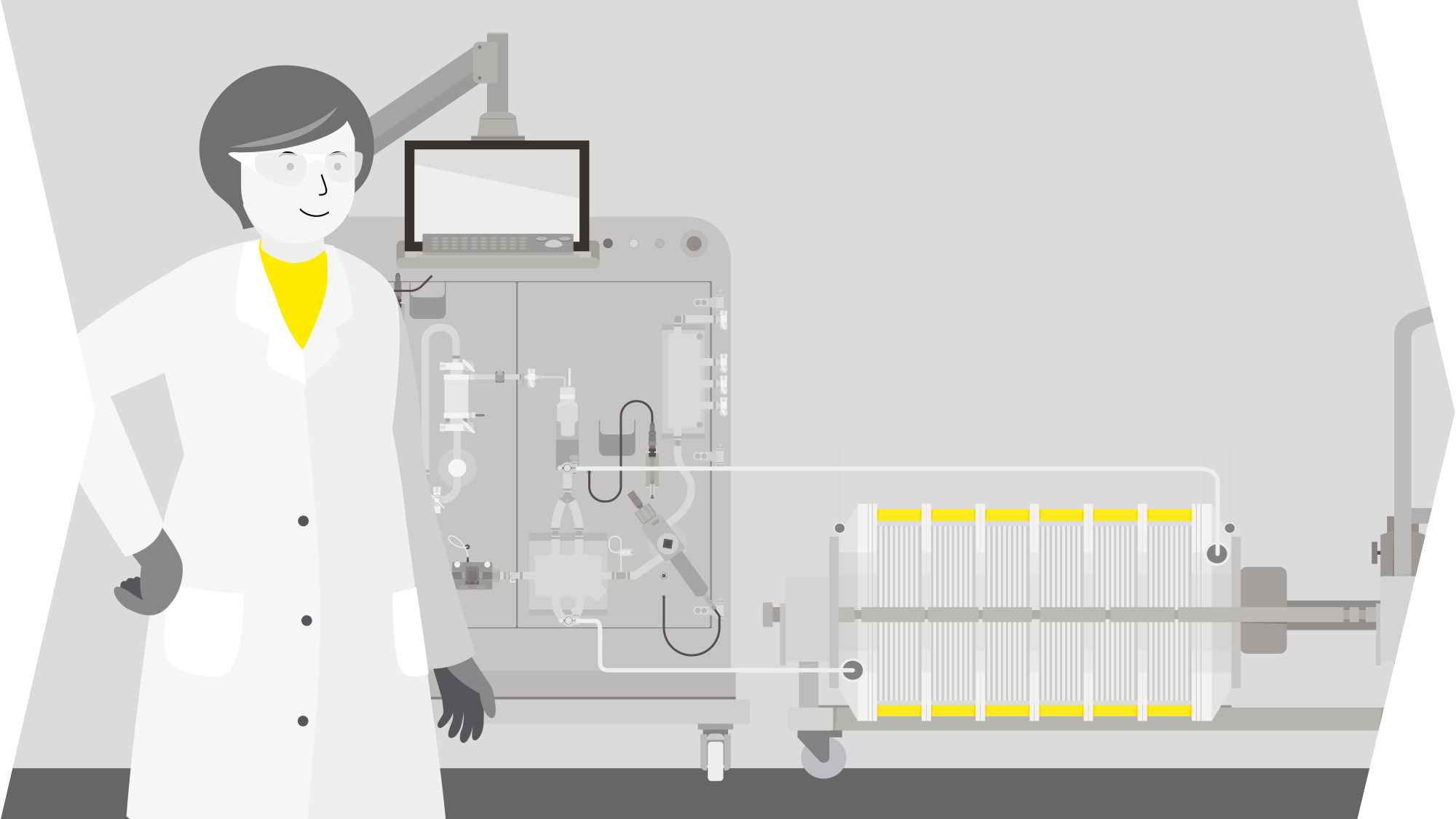
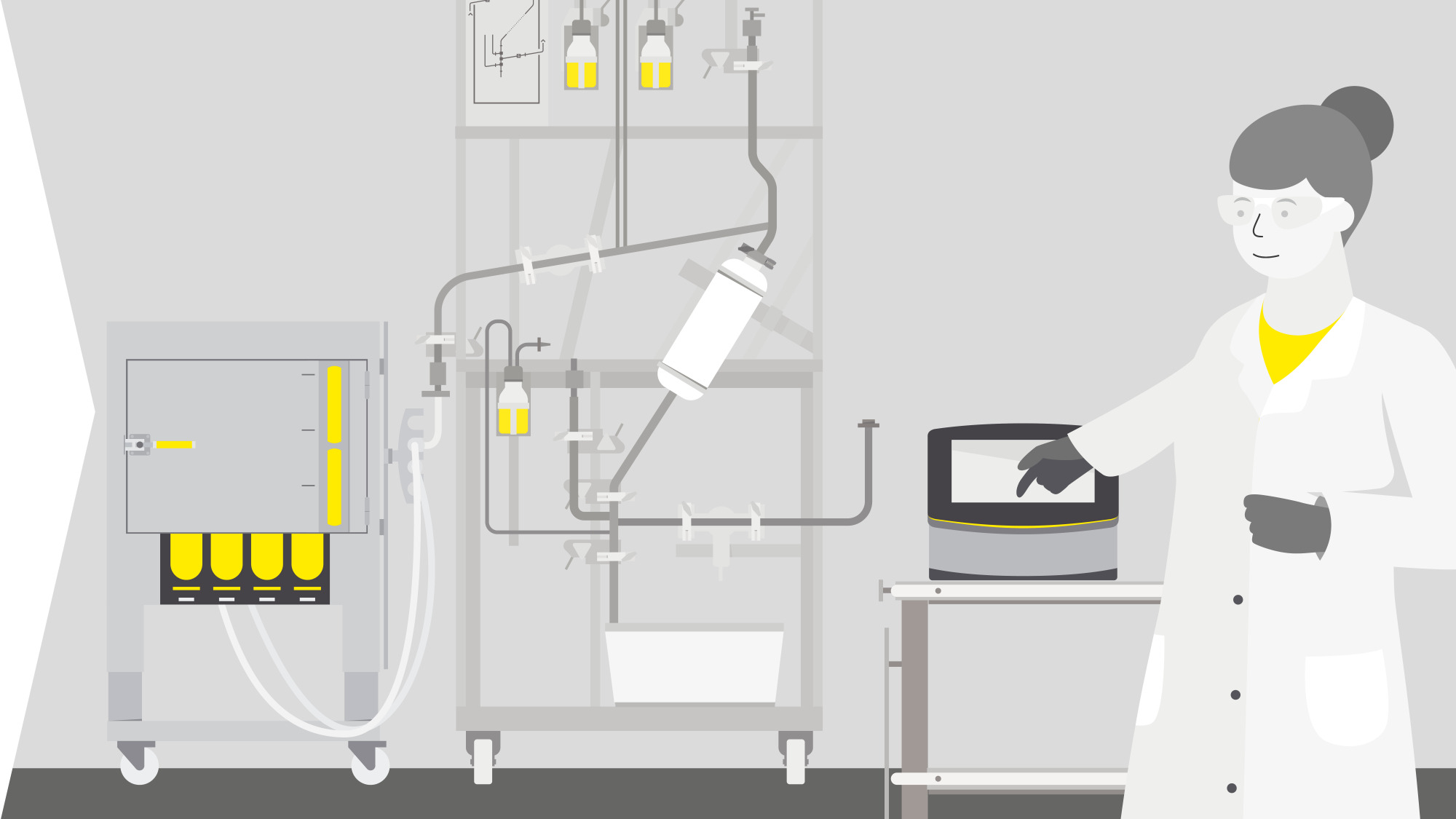
Solutions For Lentiviral Vector Production
Connected Services
Plasmid Engineering Service offers custom plasmid engineering based on a unique DNA brick approach to enable fast and precise engineering of any plasmids, even the most complex ones with a success rate above 95%
Plasmid Manufacturing Service excels in producing pDNA at high quality with superior yields and supercoiled integrity. Each plasmid DNA produced is provided with regulatory support to ensure compliance for clinical trials and commercial use.
Cornerstone Development Service is designed to support customers in establishing in-house pDNA purification processes at the required scale and that is then seamlessly transferred to each customer site.
Cell culture media Services include a array of media services including benchmarking services, media optimization, media development and media manufacturing-all supported by spent media analytics
Connected Software & Analytics
DOE software Simplified for Life Science. Ready-to-go connectors for Sartorius equipment & Biobrain Supervise to ease implementation. Backed up with a data science team to get the most out of your data.
Multivariate data analysis software that simplifies your data exploration for Process Experts. Ready-to-go connectors for Sartorius equipment and Biobrain Supervise. Backed up with a data science team to get the most out of your data.
A data acquisition, process control & monitoring system that connects Sartorius and 3rd party unit operations. A scalable system can support 1 to 32 process units to enable PD or GMP production with integration to Umetrics® for DOE or advanced process control.
Request Your Sample | Raw Materials and Consumables
To help find the right materials and consumables for your needs, request a sample from our portfolio.
More About LV Gene Therapy From Sartorius
Featured Asset
FAQs for Lentiviral Vector Gene Therapy
Lentiviruses are a family of viruses insert DNA into the host cells' genome upon transduction. They can permanently integrate genetic material into dividing and nondividing cells. Lentiviral vectors are produced from a culture of packaging cells, which scientists then transfect with plasmids. The vector-producing cells are then expanded in culture and purified.
Lentiviruses are transducing, dividing, and nondividing cells, and enable long-term expression by integrating into the cell genome. They have a large payload of 9.7 kb. Lentivirus is unique in its ability to infenct nondividing cells, and has high transduction toward targets like T-cells.
BSL2. Scientists can't perform viral clearance on lentiviruses using a filter, so single-use and closed systems are prefered. Scientists need to control raw materials and filter them to remove virus. Removing impurities like DNA and HCP is difficult because lentiviruses are large and sensitive, making downstream process challenging.