Cell Line Development With Intelligent Tools, Services, and Solutions for Your Unique Needs
A Single-Vendor Approach
When it comes to the commercialization of a biological product, effective, high-quality cell line development is essential for your success. It requires a partner that offers experience, expertise, and a comprehensive portfolio of products and services — someone who can minimize risks, optimize outcomes and increase your flexibility.
Sartorius addresses your needs at every stage of the journey to commercialization, whether you need a fully outsourced solution or select products and services to complement your in-house cell line development (CLD) capabilities.
A Flexible Portfolio for Cell Line Development and Beyond
CLD Instruments
Making the right decisions early is a key differentiator in cell line development. Enhance your selection and characterization processes by choosing high-throughput solutions that can increase lab productivity, reduce costs and shorten timelines.
CLD Services
A misstep during early-stage drug development can cause major delays and roadblocks to commercialization. Increase your chances of success by choosing a competent and experienced partner for your cell line development project.
CLD Custom Solutions
As you build your unique bioprocess, you need flexible solutions that enhance and extend your capabilities. Sartorius offers a full range of products and consulting expertise so you can get the right technologies and the right advice at exactly the right time to maximize your success.
Cell Line Development Instruments
Innovative Products and Expertise to Optimize Your Processes
When building a team and capabilities to conduct high-throughput analysis and characterization, you need a partner who offers state-of-the-art technology along with training, support, and skill development.
Sartorius’ high-throughput systems — CellCelector, Octet®, iQue® 3, and Ambr®15 —are the most popular choice of labs around the world for early identification of stable, scalable and high-titer cell lines. These innovative instruments streamline early-stage development and enable a smooth transition to manufacturing.
Increase lab productivity
Reduce costs
Shorten experimental timelines
Cell Line Development Services
Expert Outsourced Services from a Single Trusted Provider
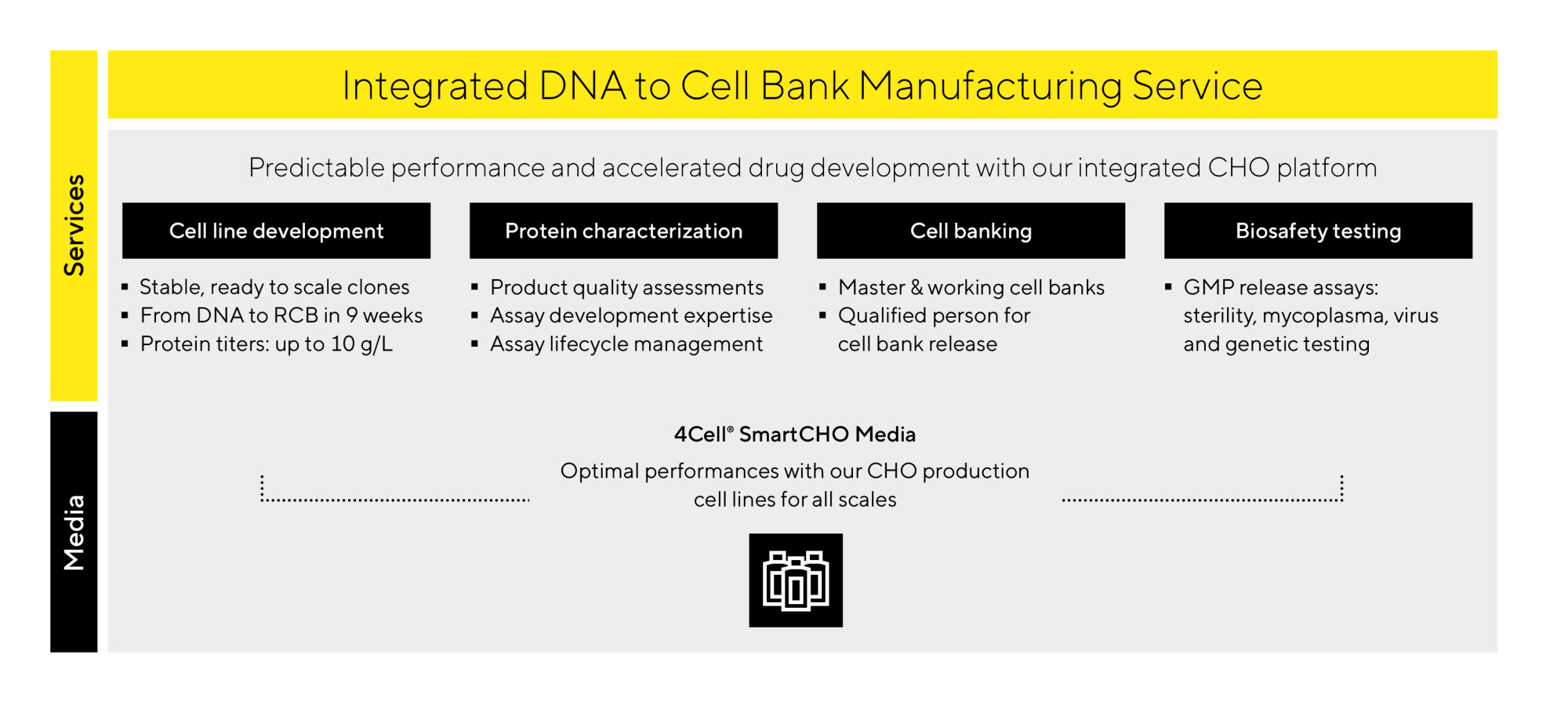
Outsourcing cell line development to an experienced service provider can ensure a smooth and efficient path to IND-readiness. Sartorius’ CHO cell line development service is built on a strong foundation of expert knowledge and cutting-edge technology. With more than 330 cell lines developed and 10 already approved for market use, the Sartorius CLD platform and technology are ideally positioned to serve as your single‑vendor solution.
We offer adaptable solutions supported by diverse capabilities, embedded expertise, and optimized data‑driven workflows to drive success toward IND submission.
- 15+ years of experience with 330+ completed cell line development projects
- 85+ molecules in clinical stages, including 10 approved for market use
- Exceptionally high titers and productivity across mAbs and complex protein formats
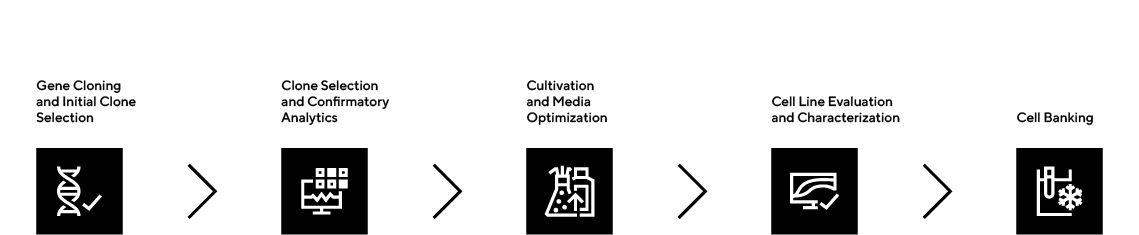
- Fast timelines: RCBs in 9 weeks; MCB manufacturing and release in 9 months
- Powered by 4Cell® SmartCHO media for optimal performance and scalability
Cell Line Development Custom Solutions
A Tailored Approach to Fit Your Process Needs
If you’re building a unique bioprocess that complements your in-house capabilities with some combination of advanced technologies, state-of-the-art instruments and experienced scientists from external providers, you benefit from a proven partner that can provide everything you need.
Sartorius offers a flexible suite of products and services and a deep bench of cell line development experts, so we can help you design a custom solution that fits your unique processes.
- Implement an adaptable approach
- Benefit from 100% flexibility
- Streamline with a single-vendor approach
Learn More From Sartorius Experts
A Guide to Accelerating CLD for Commercial Production
In this White Paper, Sartorius will provide an overview of cell line development and the associated hurdles that biotech companies should be aware of when establishing their process. Additionally, potential solutions that developers can access as part of an effective risk-management strategy and to improve their productivity are introduced.
Frequently Asked Questions
Usually both, toxicology and first-in-human clinical materials are generated using the same clonal-derived cells to ensure safety and minimize any development risks. However, cell line development with single cell cloning is time consuming and aggravated by the time needed to screen for a lead clone based on cell line stability and manufacturability. In order to achieve faster timelines, there are approaches using pools of several clones for earlier production of drug substance for regulatory filing-enabling toxicology studies, and later the final single clone will be selected for production of clinical materials.
Post-translational modification is a common event during protein production from eukaryotic cells. It includes a wide variety of chemical modifications, such as acetylation, amidation, carboxylation, phosphorylation, and glycosylation. These important chemical modifications are essential for protein folding, stability, and function.
Among these various post-translational modifications, glycosylation is the most complex one from the perspective of chemical heterogeneity of modifications because it involves the covalent attachment of sugar moieties to proteins. There are two common types of glycosylation: O-linked glycosylation and N-linked glycosylation.
The glycosylation pattern plays an important role in the biological activity of a protein, such as that of afucosylated monoclonal antibodies (mAbs).
During therapeutic antibody production via mammalian cells, such as Chinese hamster ovary (CHO) cells, N-linked glycosylation is commonly observed. For the IgG1 type of antibodies, there is a conserved site in the heavy chain Fc region for glycosylation. During the post-translational modification, multiple sugar moieties (e.g., N-acetylglucosamine (GlcNAc), mannose, fucose, galactose) can be assembled with complex combinations to form a variety of glycosylation patterns. Advances have been made primarily in upstream processing, including mammalian cell line engineering, to yield more predictably glycosylated mAbs and the addition of media supplements during fermentation to manipulate the metabolic pathways involved in glycosylation. Another strategy is the implementation of novel downstream technologies, such as the use of Fcγ-based affinity ligands for the separation of mAb glycovariants.
It is common in cell line development to get a certain percentage of clones that unstable, meaning that they lose productivity over time due to a loss of gene copies or gene silencing effects. Depending on the applied technology the proportion of unstable clones can be up to 50 % or higher. However, with mature technologies such as Sartorius’ Cellca Cell Line Development Technology the percentage of stable clones can be 80 % or more.
Nearly all cellular systems are heterogeneous, contributing to specialized functionality and improved survival. A profound comprehension of single cell heterogeneity on genomic, epigenomic, transcriptomic and proteomic level is critical for understanding its impact on the functioning of organism in both healthy and diseased condition.
Most of our current knowledge about different cell and tissue types comes from bulk assays though, analyzing hundreds to millions of cells together, which may highly underestimate the true spectrum of cellular heterogeneity.
Modern technologies allow the analysis on single cell level therefore taking a huge step forward in the understanding of cellular heterogeneity and its implications:
- Single cell heterogeneity analysis can help to clarify developmental pathways, e.g., how stem cells make their fate decision, contributing to the development and maintenance of differentiated tissue. This is not only important for understanding malignancies caused by missing or misdirected differentiation but especially for the field of regenerative medicine.
- In order to analyze the cellular heterogeneity on single cell level e.g., by whole genome amplification (WGA), SNP detection or sequencing target cells have to be selected and isolated in high purity from a heterogeneous background. The CellCelector™ is the perfect tool for the automated identification and isolation of 100 % pure individual single cells offering many advantages over other methods.
- Target cells are detected and selected for isolation based on microscopic imaging. This enables target cell selection utilizing fluorescent markers but also through distinct morphological features a target cell might express and that is visible in microscopy. This allows label free detection of target cells independently of fluorescent markers. Furthermore, a live image during isolation as well as automated recording of before and after picking images of each isolated single cell create a comprehensive documentation of the picking process ensuring that only target cells have been picked without contamination of non-target cells. The fast isolation process is extremely gentle resulting in high cell integrity (up to 100 % outgrowth rate in cloning applications) and outstanding transfer efficiency of up to 100%.