Watch Now: Challenges and Solutions in Developing CAR-T Cell Therapies
Chimeric antigen receptor (CAR) T-cell is a significant breakthrough in the battle against cancer and a revolutionary step towards individualized treatment. While CAR-T cell therapy has shown impressive clinical outcomes in specific groups of B-cell leukemia or lymphoma, there are several obstacles that hinder the effectiveness of CAR-T cells in treating solid tumors.
This rapidly growing field faces several challenges, from basic research and early-stage clinical trials to Investigational New Drug (IND) applications and final product release. These challenges include comprehensive screening of the product as well as quality control testing to ensure patient safety.
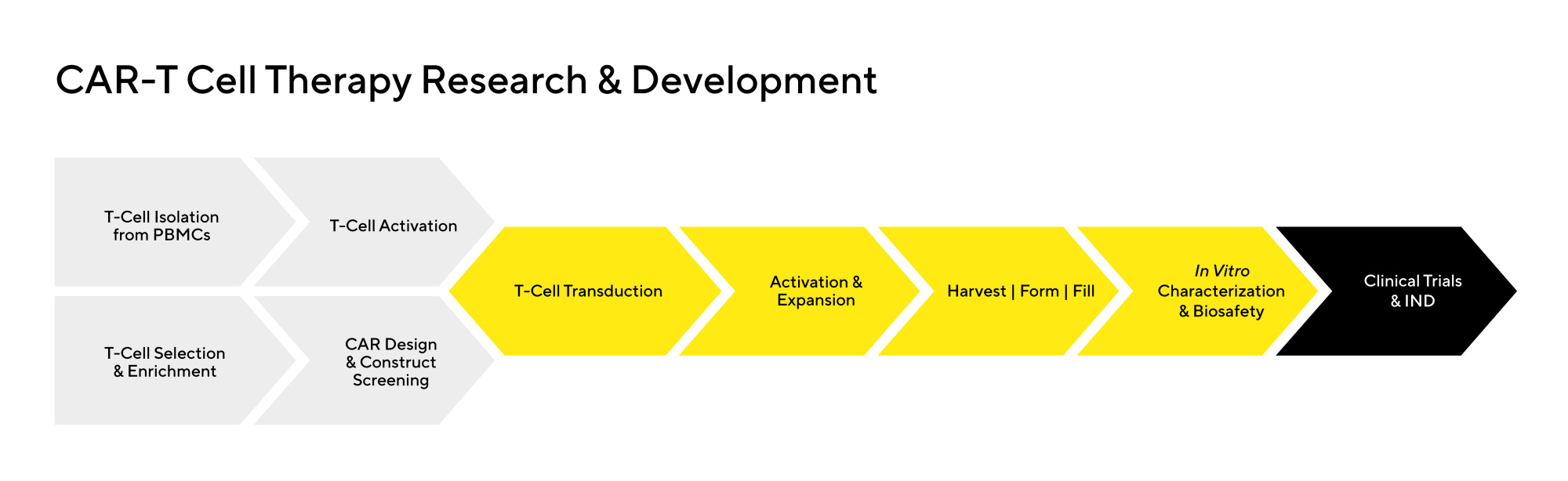
Additional challenges in research and development of effective CAR-T cell therapies include:
- Target selection: Identifying the right target antigen is crucial for the success of CAR-T therapy. The target antigen should be expressed on cancer cells but not on healthy cells to avoid off-target toxicity.
- Manufacturing: CAR-T cells are manufactured by genetically modifying a patient's T cells, which is a complex and time-consuming process. The manufacturing process needs to be standardized and scalable to produce enough CAR-T cells for widespread use.
- Safety: CAR-T cells can cause severe side effects, including cytokine release syndrome (CRS) and neurotoxicity. Developing strategies to mitigate these side effects is critical for the safety of patients.
- Efficacy: CAR-T therapy has shown remarkable clinical responses in certain types of blood cancers, but its efficacy in solid tumors is limited. Developing strategies to improve the efficacy of CAR-T therapy in solid tumors is an active area of research.
- Cost: CAR-T therapy is expensive, and the high cost of treatment is a significant barrier to its widespread use. Developing cost-effective manufacturing processes and reimbursement strategies is critical to making CAR-T therapy accessible to more patients.
Watch the Video: Challenges and Solutions in Developing CAR-T Cell The...
Watch this video to learn more about the steps and overcoming challenges in bringing a new CAR-T therapy to patients.
Five Things You Will Learn About
- Challenges in a CAR-T development workflow
- Importance of developing critical quality attributes (CQAs)
- Ensuring consistency of results and controlling variability
- Screening for high-affinity CAR constructs using automated, high-throughput instruments
- Information-rich assays for T-cell phenotyping, cytokine analysis, ligand affinity, and T-cell killing